Results
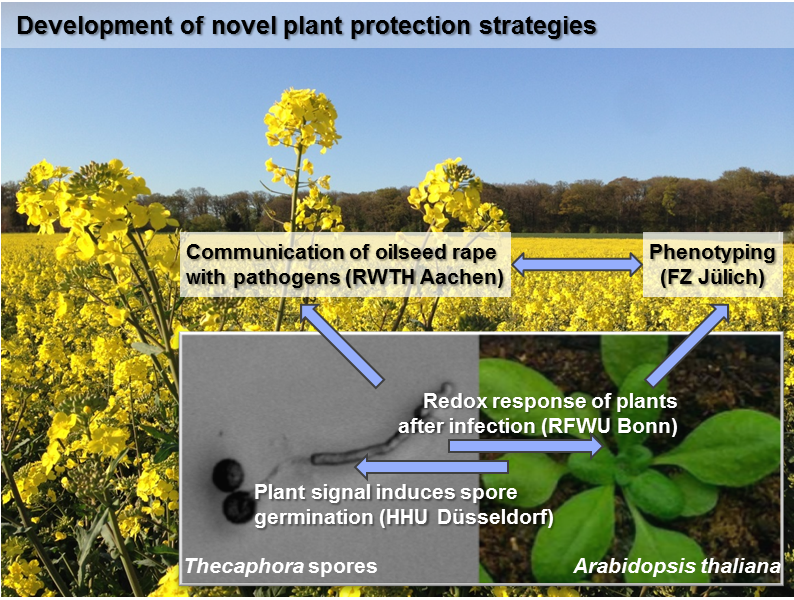
Rapeseed (Brassica napus, Bn) is an economically important crop plant which is not only used for food production but also as a renewable resource for fuel production. The fungal plant pathogen Verticillium longisporum (Vl) is one of the major threads of rapeseed, which may cause yield losses of up to 50%. The project PlaMint worked towards a novel plant protection strategy for Bn against this pathogen by exploiting different features of biotic interactions. PlaMint aimed at bridging the gap between basic science and agriculture by transferring knowledge obtained for the model plant Arabidopsis thaliana (At) to the bio-economically relevant crop plant Bn. To gain a deeper insight into plant-pathogen interactions, two fungi with initially similar infection strategies were compared. Both the biotrophic smut fungus Thecaphora thlaspeos (Tt) and the hemi-biotrophic pathogen Vl can infect Brassicaceae; however, development of the disease proceeds very different. Towards a proof-of-concept a combination of dynamic live cell imaging, molecular genetics, high-throughput phenotyping and genomics was used. We have established the Tt-Brassicaceae system such that it can be used to study plant–microbe interaction. A key step is the successful sequencing of the fungal genome and initial characterization of fungal virulence factors. We also developed fungal reporter lines with expression of different fluorescent reporter proteins which can be used for further plant-pathogen interaction studies in the future. Unexpectedly, initial tests with Bn plants that show opposite infection phenotypes with Vl showed no difference when infected with Tt. Therefore, further phenotyping experiments and the molecular characterization focused predominantly on the interaction of Bn and At with Vl. In gene expression studies, the Vl tolerant line R53 showed that a large number of differentially expressed genes correlated with cellular redox homeostasis compared to two susceptible lines. Further evidence for the involvement of redox processes resulted from in vivo studies of At expressing redox-sensitive reporter proteins. Especially in a mutant background lacking cytosolic glutathione reductase the sensors reliably report elicitor-induced oxidative changes. The infection with Vl, however, triggered complete loss of fluorescence within 3 days in a mechanism that could not be explored yet. With the establishment of the interaction of At with the model smut fungus Tt as an alternative experimental system and several Tt strains with expression of fluorescent reporter proteins PlaMint build a platform that can be used for further development of novel plant protection strategies as one the key goals of the Bioeconomy Science Center for a sustainable bioeconomy.
Participating Core Groups
Prof. Dr. Andreas Meyer, INRES - Chemical Signalling, University of Bonn
Dr. Vera Göhre and Prof. Dr. Michael Feldbrügge, Institute of Microbiology, Heinrich-Heine-University Düsseldorf
PD Dr. Ulrich Schaffrath Institute of Biology III (Plant Physiology), RWTH Aachen and Prof. Dr. Björn Usadel, Institute of Biology I (Botany), RWTH Aachen/FZ Jülich
Dr. Robert Koller and Prof. Dr. Ulrich Schurr, Institute of Bio- and Geosciences IBG-2: Plant Sciences, Forschungszentrum Jülich GmbH
(Coordinator)
Prof. Dr. Andreas Meyer
INRES – Chemical Signalling
University of Bonn
Friedrich-Ebert-Allee 144
53113 Bonn
phone: +49 (0) 228 - 73 60353
e-mail: andreas.meyer@uni-bonn.de
Project duration
01.11.2015 – 31.10.2018
Funding
PlaMint is part of the NRW-Strategieprojekt BioSC and thus funded by the Ministry of Innovation, Science and Research of the German State of North Rhine-Westphalia.
Publikationen
Bosch, K, Frantzeskakis, L, Vranes, M, Kamper, J, Schipper, K and Gohre, V (2016). Genetic manipulation of the plant pathogen ustilago maydis to study fungal biology and plant microbe interactions. J Vis Exp(115).
Frantzeskakis, L, Courville, KJ, Plucker, L, Kellner, R, Kruse, J, Brachmann, A, Feldbrugge, M and Gohre, V (2017). The plant-dependent life cycle of thecaphora thlaspeos: A smut fungus adapted to brassicaceae. Mol Plant Microbe Interact: Mpmi08160164r.
Kellner, R and Göhre, V (2017). Thecaphora thlaspeos–ein Brandpilz spezialisiert auf Modellpflanzen. BIOspektrum 23(5): 498-500.