Main focus of the development area “technology advancement” (CG Schwaneberg, CG Pich and CG Gohlke) is the development of microgel release containers for pesticides and herbicides, the identification of leaf binding anchor peptides, and the understanding of pesticide release kinetics as well as peptide leaf interactions. Microgels are 3D polymer networks that are swollen by water and show a hydrodynamic diameter between 100 nm to 100 µm. There are various ways to synthesize microgels, but the most commonly used method is precipitation polymerization. These smart materials show a fast response to outer stimuli and are favorable candidates in wide application fields, for example nutrition delivery or drug release on demand. We designed microgels with hydrophobic or hydrophilic domains to load and release different active molecules (pesticides and herbicides). By applying all-atom molecular dynamics simulations, we gained insight not only in the thermo-responsiveness of the smallest microgel building blocks, but also in the growth process of microgels. We constructed atomistic models of microgels that show the same characteristics observed in experiments. These models were used to describe the uptake and release processes of fungicides and herbicides and to tailor and optimize this process.
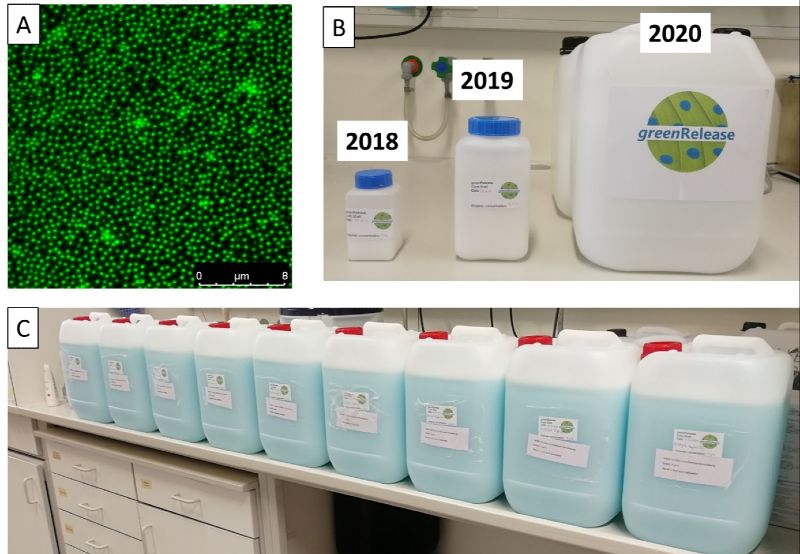
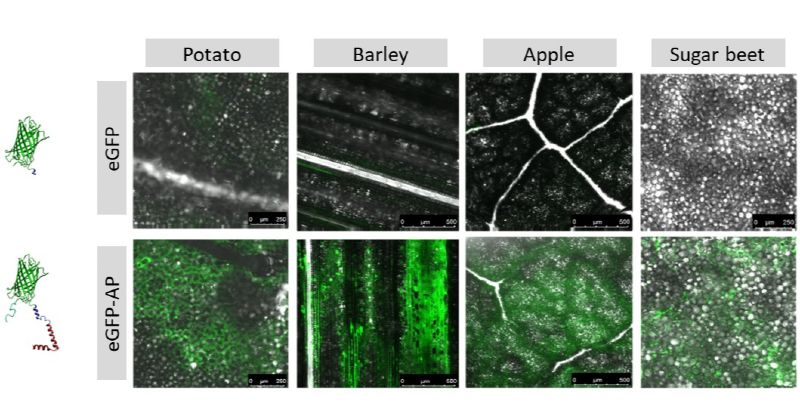
The microgel containers are decorated with anchor peptides (Fig.1 A), acting as adhesion promoters for plant surfaces in order to enable selective and strong binding of the pesticides loaded microgels and to minimize run off of active compounds from plants. Anchor peptides are small molecules with sizes ranging from 10 to 100 amino acids. Anchor peptides bind to surfaces at ambient temperature by simple spray applications. The binding strength of an anchor peptide to a given surface can be increased by protein engineering. Within the greenRelease project, we identified strong binding anchor peptides for the leaves of all selected target plants (apple, sugar beet, potato, barley; Fig. 2). A directed evolution protocol for anchor peptides which yields >2 amino acid substitutions per variant was developed in order to further increase the rain fastness of anchor peptides. To improve the rain-fastness of the applied microgel container, understanding the adsorption of the anchor peptides to the leaf surface is the key.
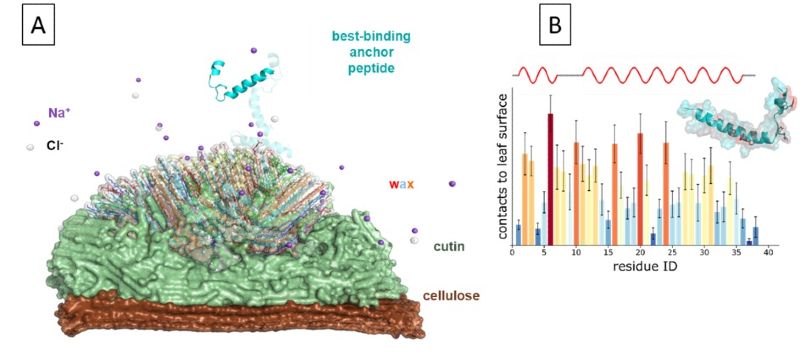
Therefore, we first generated novel atomistic models of different leaf surfaces to probe the adhesion properties of the anchor peptides. Based on the experimentally determined leaf wax composition, we developed a three-layer model of an apple tree leaf surface consisting of a rigid cellulose layer covered with a polyester cutin matrix and leaf wax on top. The model was used in consecutive all‑atom molecular dynamics simulations to probe the anchor peptides for hotspots that are essential for adhesion and serve as guidance for future tailoring of the adhesion properties (Fig. 3).
Within the course of the project we developed a production strategy that allowed us to produce and deliver 120 L of the greenRelease formulation for field trials (Fig.1 B,C) that were performed in 2020 by the agricultural chamber of North Rhine-Westphalia.
Prof. U. Schwaneberg
Dr. F. Jakob
Chair of Biotechnology
RWTH Aachen University & DWI – Leibniz-Institute for Interactive Materials
Prof. A. Pich
Institute of Technical and Macromolecular Chemistry
RWTH Aachen University
Prof. H. Gohlke
Computational Pharmaceutical Chemistry and Molecular Bioinformatics
HHU Düsseldorf