
Natural products, which are produced as secondary metabolites by organisms from all kingdoms of life, represent a rich source of bioactive compounds. They can be applied in the agricultural and the pharma sector or serve as chemical frameworks for developing new agrochemicals and pharmaceuticals where new agents are urgently required to meet food and health demands of modern societies.Reinforcing the change of paradigm from synthetic chemistry to sustainable microbial production, the CombiCom project has thus exploited natural product biosynthetic pathways to deliver such bioactive natural products and structurally derived compounds. Addressing potential market introduction, this was accompanied by the assessment of relevant bioactivities and an evaluation of the social acceptance of synthetic biology associated products. The consortium of nine BioSC Core Groups collaboratively developed the project over the years 2017-2020.
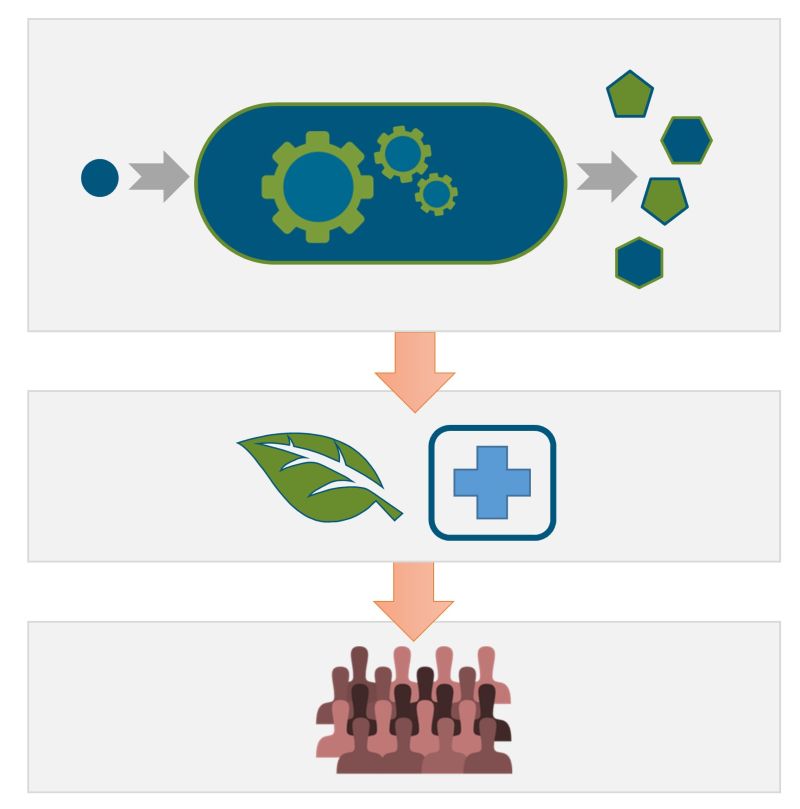
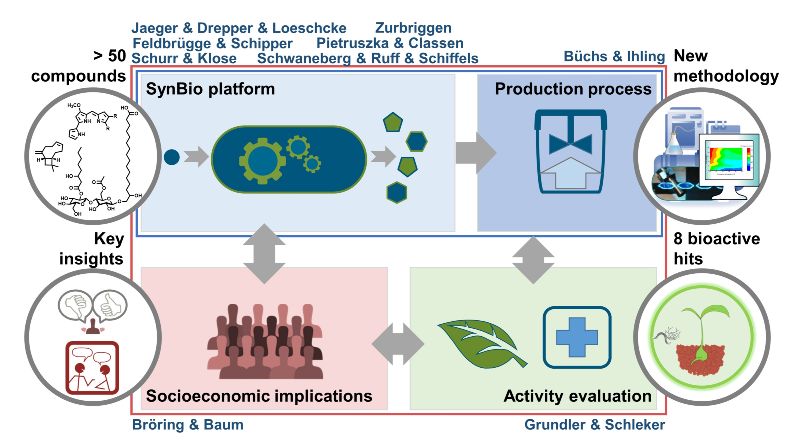
We established a “synthetic biology platform” with the bacteria Rhodobacter capsulatus and Pseudomonas putida, the fungus Ustilago maydis and Chlorellaceae green algae to exploit specific features of non-photosynthetic and photosynthetic organisms. They were equipped with genetic modules to produce diverse natural terpenoids, prodiginine pyrrole alkaloids and glycolipids – as well as derivatives thereof. To this end, we developed and combined smart tools, especially focusing on genomic integration of genes, multi-gene expression, (opto)genetic fine-control of gene expression and pathway-specific precursor biosynthesis. Their application together with chemical precursor modification and enzyme engineering enabled the production of over 50 biomolecules, which were hitherto difficult to obtain. For especially promising microbial strains as well as representatives of the microalgal Chlorellaceae group, we developed tailored bioprocess approaches for microbe-based compound production and extraction procedures.
Here, for example, newly established online monitoring methodology enabled following prodigiosin production by P. putida in real-time during cultivation. Key insights into growth and production characteristics directed bioreactor-based pilot production.
Terpenoid production was particularly successful with R. capsulatus and U. maydis, the latter also producing high levels of glycolipids, while diverse prodiginine structures were obtained using P. putida as a chassis and combining synthetic biology and synthetic chemistry. Similarly, for the green microalga Parachlorella kessleri, a new methodology of cell-wall permeabilisation was developed to facilitate product extraction and molecular manipulation.
Gathered knowledge about the strains’ individual and unique capacities shall contribute to transfer each microbial chassis along with a designated toolkit to a next level of applicability in biotechnology for natural compound production.
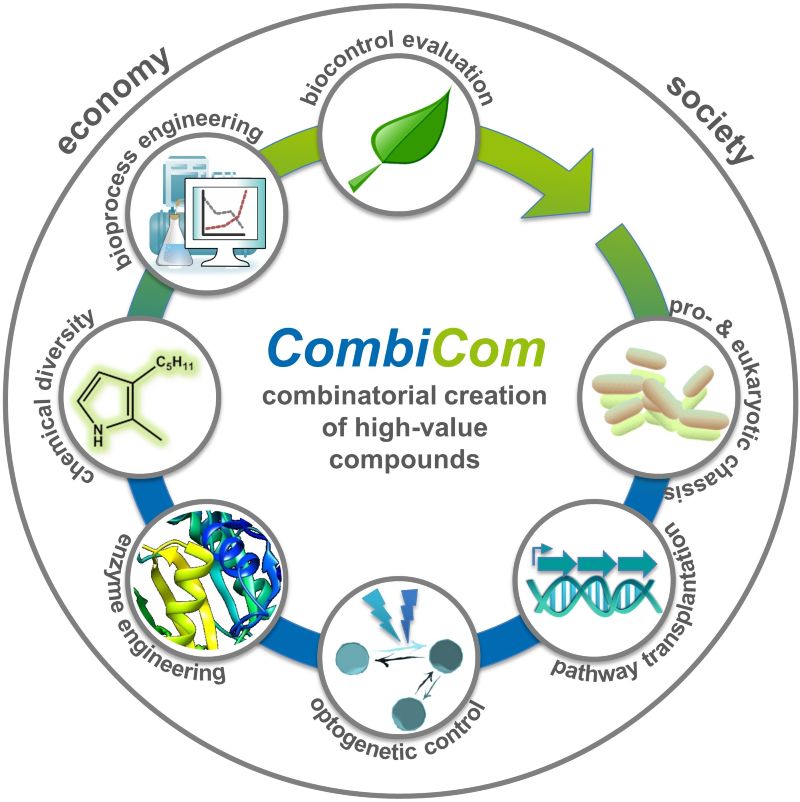
We further directly subjected CombiCom compounds to investigation of their plant protection and pharmacological potential. We identified several promising bioactive ‘hits’ that have the potential to be developed into novel antiphytopathogenic agents: For example, among a set of terpenoids, nootkatone was identified to inhibit the plant parasitic nematode Heterodera schachtii by activating plant defense and β-caryophyllene was found to have specific activity against plant pathogenic fungi like Sclerotinia sclerotiorum. Specific prodiginines showed pronounced activity against fungi, nematodes as well as cytostatic activity in breast cancer cells. Finally, we explored key barriers and success factors for the commercialisation of such products. Here, we highlighted how understanding of “drivers for the emergence of synthetic biology” differed across groups of stakeholders within an “invention ecosystem” that is emerging – focusing on the case of the Heinrich-Heine-University Düsseldorf. A further key insight regarding the acceptance of new technologies, which is a crucial determinant for successful market introduction, pertained to the importance of positively establishing potential benefits to consumers – including by engaging certain actors along the value chain and across the emerging invention ecosystem.
The FocusLab project CombiCom has thus yielded important results boosting the microbial production of known and new high-value bioproducts.

Dr. Anita Loeschcke
Institute of Molecular Enzyme Technology of the Heinrich-Heine-University Düsseldorf
located at Forschungszentrum Jülich, Geb. 15.8
Stetternicher Forst
52426 Juelich
Email: a.loeschcke@fz-juelich.de
01.05.2017 – 31.12.2020
CombiCom is part of the NRW-Strategieprojekt BioSC and thus funded by the Ministry of Culture and Science of the German State of North Rhine-Westphalia.