
The FocusLab Bio² aimed at the development of a competitive biorefinery process for the production of bio-based surfactants. Rhamnolipids (RLs) and mannosylerythritol lipids (MELs) were produced by recombinant strains of Pseudomonas putida and Ustilago maydis, respectively. The project combined strain optimization with process development and in situ product recovery approaches to account for the complex process requirements. A socioeconomic evaluation via life cycle assessment guided the process design up to the evaluation in technical scale. By opening up new raw material sources, increasing yields and providing new analytical techniques, the project helped to enhance biosurfactant competitiveness on the market.
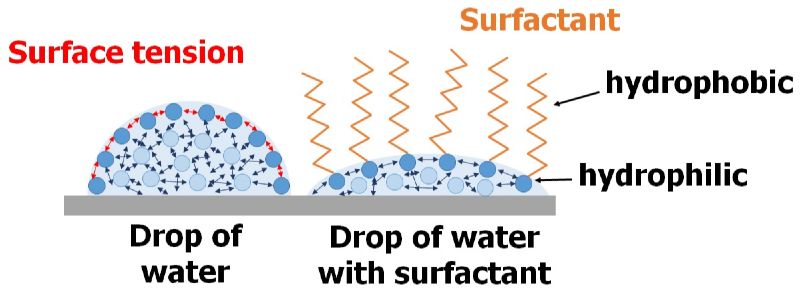
Completely bio-based surfactants („biosurfactants“) represent a sustainable alternative to surfactants produced from crude oil. (Bio)surfactants reduce the surface tension of liquids or the interfacial tension of dispersions (Figure 1). This enables a wide range of applications. Consequently, surfactants are used as detergents in cleaning agents, as foaming agents in firefighting, and as emulsifiers in the food industry. Biosurfactants are of microbial origin and can be produced from renewable raw materials.
Production costs for biosurfactants are higher compared to surfactants produced from crude oil. This is caused by the use of expensive raw materials and by low yields and production efficiencies of the microorganisms. Furthermore, the use of sugars or oils as raw materials competes with food production and has to be evaluated critically. Additionally, process control in classical bioreactors is hampered by the biosurfactant’s inherent property of foam formation. Although foaming can be reduced by adding antifoaming agents, this impedes the purification process.
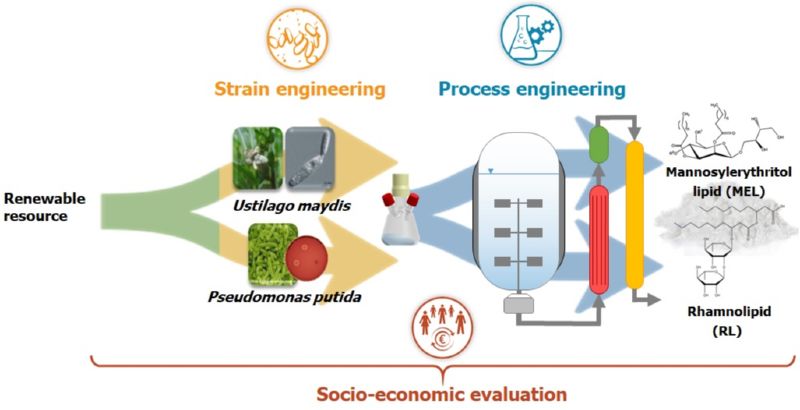
The aim of Bio2 was to address these challenges in a holistic approach along the entire process chain. Therefore, strain and process engineering were considered simultaneously and process stages were interconnected. The work was accompanied by a socio-economic analysis to evaluate and improve the resulting process in terms of ecological, economic and social aspects. Rhamnolipids produced by Pseudomonas putida and mannosylerithrytollipids (MEL) produced by Ustilago maydis were selected as examples of biosurfactants. Fig. 2 shows the basic project structure.
The holistic approach could only be implemented in a highly interdisciplinary team. Accordingly, natural scientists and engineers from different disciplines worked closely together within the project.
Growth on renewable resources
For Pseudomonas putida, the substrate spectrum was extended to xylose and ethanol. Xylose is an important constituent of lignocellulose. Consequently, investigation of rhamnolipid production for example from beech wood hydrolysate will be possible. This way, a direct link to the FocusLab AP³ was created. Expansion of the substrate spectrum towards ethanol opens up further options to use alternative carbon sources.
For Ustilago maydis, metabolization of polygalacturonic acid was achieved by the overexpression of heterologous polygalacturonases. This leads the way to using substrates containing pectin, such as sugar beet pulp (SBP). SBP is a by-product of sugar beet processing. Also, it was evaluated if the biomass remaining after the production process can be used as fermentation substrate to utilize remaining waste streams.
Strain development to increase product quantities
For P. putida, numerous promoter systems for the expression of the rhamnolipid biosynthesis genes were adapted. These increase the genetic stability and allow the optimization of the production, for example by adjusting the induction timepoint. Additionally, the genome was reduced to reduce energy-intensive applications such as the formation of flagella. A specially developed high-throughput assay in microtiter plate format considerably simplifies the comparative analysis of glycolipid producers.
When using renewable raw materials, the nitrogen content cannot be controlled at will. To reduce the nitrogen dependency of the MEL production in U. maydis, a 2A peptide strategy for the artificial regulation of the biosynthesis steps was established. Furthermore, important steps for the production of pure MEL-D production could be achieved by eliminating side products such as ustilaginic acid and itaconic acid.
Holistic process development
A new concept for aerobic cultivation - membrane gassing - was developed within the project and a patent application was filed. The integration of the corresponding modules into existing bioreactors enables almost bubble-free aeration of the bioreactor and nearly completely avoids foam formation in the process. The interconnection of the membrane module with a filtration membrane for cell retention also provides the basis for in-situ product separation.
For purification of the glycolipids from the fermentation broth, different process alternatives were evaluated, which achieve high product purity combined with high recovery and thus low product losses. Pre-selected separation operations were examined experimentally and model-based screening experiments were performed. Afterwards, purification processes were established by interconnecting the examined separation operations. The corresponding process chains were evaluated for different substrates from a socio-economic point of view. The accompanying evaluation of various environmental indicators as part of a life cycle assessment guided the process optimization.
The holistic process approach was successfully put into practice during the project. This is expressed, among other things, in a joint publication on the project's rhamnolipid part.
Project leader

Dr. Nina Ihling
AVT - Aachener Verfahrenstechnik
RWTH Aachen University
Forckenbeckstraße 51
52074 Aachen, Germany
E-mail: Nina.Ihling@avt.rwth-aachen.de
Phone: +49 241 80-47868
Fax: +49 241 80-22570
Funding Period
01.05.2017 – 31.12.2020
Funding
Bio² is part of the NRW-Strategieprojekt BioSC and thus funded by the Ministry of Culture and Science of the German State of North Rhine-Westphalia.