Lignocellulosic material, i.e. plant cell walls constitute the most abundant non-fossil plant-based resource on earth. Therefore, it is an ideal candidate to help to replace fossil resources. To increase economic figures and minimize the ecological footprint of the process, a full valorisation of lignocellulose is of major importance.

Plant lignocellulosic material consists of three main polymers: cellulose, a crystalline polysaccharide consisting of glucose, the polyaromatic amorphous compound lignin, which lends solidity and brittleness to the cell walls, and the amorphous polysaccharides hemicelluloses which, due to the varying composition of hexoses and pentoses, are random in structure. Hemicelluloses connect lignin with cellulose, rendering the cell wall a highly complex structure. The high complexity and variability in composition is the reason why the fractionation into the different compounds is a challenge, a fact referred to as recalcitrance of lignocellulose. Specific factors causing recalcitrance are, for example, the degree of crystallinity of cellulose; the lignin content, structure and ratio of subunits and the density and number of linkages between subunits and with associated polysaccharides; and possibly hemicellulose sugar substituents as well as the number of acetyl moieties. The recalcitrance of lignocellulose not only differs between plant species and developmental stages but also depends on the chosen pre-treatment process since different chemicals and process conditions result is varying quantities and qualities of the fractionated compounds. However, the successful recycling of water, solvents and catalysts used in the process is equally important as the full fractionation of the biomass.
Within AP3, we aimed at implementing the OrganoCat pretreatment to approach these goals, i.e. full valorisation of all three polymers of lignocellulose and recycling of the chemicals applied. Using OrganoCat as the default process, we analysed 10 different lignocellulosic plant residues aiming at identifying common features that affect recalcitrance of the biomass specifically for this process. The assessed aspects were how well the pulp can be accessed by enzymatic catalysis after OrganoCat pretreatment, how efficient hemicellulose hydrolysis is, and the extracted lignin yield and quality.
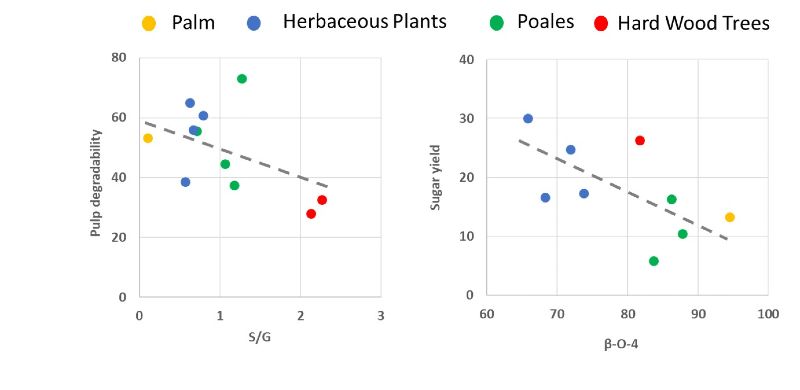
Our results show that, indeed, the ratio between lignin subunits sinapyl alcohol (S) and coniferyl alcohol G (S/G ratio) serves to a certain extent as an indicator of recalcitrance during OrganoCat pretreatment (Fig. 2). Also, polysaccharide-specific parameters correlate with recalcitrance during the OrganoCat process. In Poaceae (grasses) for example, a low amount of arabinosyl residues indicates reduced recalcitrance, possibly due to a lower number of ester bonds to the lignin. Finally, it was shown that pulp degradability after OrganoCat pre-treatment negatively correlates with the degree of xylan acetylation and lignin content in the organic phase. Thus, it is possible to predict OrganoCat product yields and qualities of all main cell wall polymers based on certain features of the lignocellulose biomass. Particularly the S/G ratio and xylan-O-acetylation of hemicelluloses are important variable features. Especially energy grasses and straw are highly predictable with respect to effective pretreatment and fractionation with the OrganoCat technology.

Within AP3, one goal is to optimize the fractionation which, as shown above, depends on the lignocellulose composition. To avoid land use conflicts, we aim at producing the plant biomass for such biorefinery concepts on marginal soils fertilized by recycled and residue-based fertilizers.
To identify the effect of specific cultivation practices on the cell wall composition and efficiency of OrganoCat pretreatment we have chosen genetically different accessions of the perennial forb Sida hermaphrodita (Fig. 3) and the perennial grass Brachypodium distachyon as model plants. The plant nutrient nitrogen was supplied in distinct chemical forms that are commonly found in mineral as well as organic fertilizers. Biomass production, cell wall composition and the success of OrganoCat pretreatment were evaluated. Sida hermaphrodita seedlings belonging to genetically different accessions responded differently to N, provided in the form of nitrate (NO3-) or ammonium (NH4+) under semi-controlled greenhouse conditions (Fig. 4).
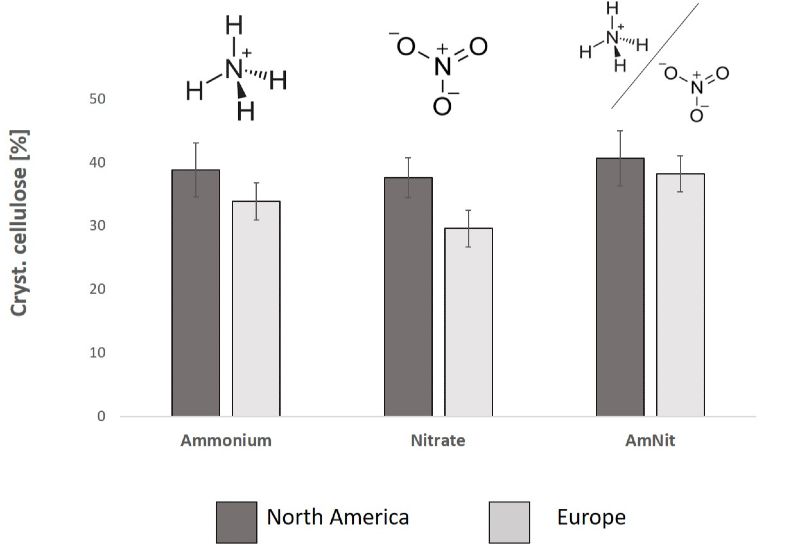
Under highly controlled laboratory conditions, the cell wall composition of Brachypodium was particularly modified during early growth stages, indicating the possibility to manipulate the plant cell wall composition via nutritional regimes. Our first results confirm literature reports that nitrogen forms affect cell wall composition in a plant species and developmental stage specific manner. Also, in residual straw, lignin content was shown to be modified in response to nitrogen fertilization.
To be able to stir cell wall compositions for successful pretreatment and to obtain optimal biomass yields of the chosen model plants, we now aim at studying both plants under controlled conditions. We expect nitrate to modify cellulose and lignin deposition while ammonium might affect the concentration of mixed linkage glucans. In addition, we will look into the chemical structure of pectins and hemicelluloses with respect to alterations in xylan substitution and esterification of homogalacturonan. Plant material with modified cell wall compositions will then be analysed regarding their processability in OrganoCat pretreatment.
Prof. U. Schurr
Dr. S. Schrey
Institute for Bio- and Geosciences: Plant Sciences
Forschungszentrum Jülich GmbH
Prof. P. Westhoff
Dr. E. Pestsova
Institute for Developmental and Molecular Biology of Plants
Heinrich Heine University Düsseldorf
Prof. B. Usadel
Dr. H. Klose
Institute of Bio- and Geosciences - Plant Sciences
Forschungszentrum Jülich
Prof. M. Pauly
Dr. M. Dama
Institute for Plant Cell Biology and Biotechnology
Heinrich Heine University Düsseldorf