Summary
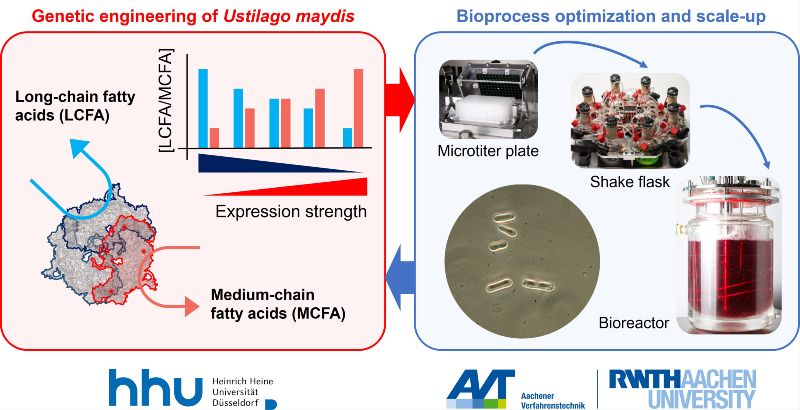
Medium-chain fatty acids (MCFAs, C6-C12) are of increasing importance for various industrial applications from personal care products to lubricants and specialty polymers. In the spirit of bioeconomy, a sustainable, environment-friendly microbial production would be highly advantageous. However, the current production hosts have certain disadvantages, including limitations in the use of sustainable substrates like waste streams and the problem of product toxicity. To tackle these challenges, FamoUs explores a sustainable, scalable approach to MCFA production in producttolerant strains from Ustilago maydis from a sugar-rich industrial waste stream. Targeted strain engineering will be tightly integrated with modern bioprocess development . To enable the switch from native long-chain fatty acids (LCFA) to synthetic MCFA biosynthesis, we will make use of U. maydis’ unique fatty-acid synthase (FAS) complex encompassing two single-chain fatty-acid synthases (Fas1 and Fas2). By addressing chain-length control in Fas2 that is upregulated during lipid production, we will uncouple growth and MCFA production while keeping the vital housekeeping LCFA production at a basal level via Fas1. Mutant variants will enable to test MCFA production capacities and to tailor chain length. On the process side, we will implement high-throughput adaptive laboratory evolution (ALE) in the µTOM platform to evolve robust tolerance against elevated concentrations of MCFAs. Genome sequences of top performers will be interpreted jointly for rational genetic engineering. To increase sustainability, we will characterize a sugar- and organic acid-rich waste stream from Zentis as fermentation feedstock and quantify how individual organic acids as well as the native mixture influences chain-length distributions. Finally, the most promising strain-medium-combination is scaled up to bioreactors. Together, we will deliver an integrated strain-and-process toolbox that (i) produces MCFA from a real industrial waste stream, (ii) offers clear tailoring options for chain-length control through genetics and cultivation conditions, and (iii) validates performance at bioreactor scale. FamoUs exemplifies BioSC’s interdisciplinary, integrated-biorefinery focus and positions U. maydis as a versatile chassis for circular, value-added lipid biomanufacturing.
Dr.-Ing. Valentin Steier
AVT - Biochemical Engineering
RWTH Aachen University
email: valentin.steier[at]avt.rwth-aachen.de
Dr.-Ing. Valentin Steier, Prof. Dr.-Ing. Jørgen Magnus, AVT - Biochemical Engineering (AVT.BioVT), RWTH Aachen University
Dr. Kerstin Schipper, Prof. Dr. Michael Feldbrügge, Institute of Microbiology, Heinrich Heine University Düsseldorf
01.11.2025 - 31.12.2026
FamoUs is part of the NRW-Strategieprojekt BioSC and thus funded by the Ministry of Culture and Science of the German State of North Rhine-Westphalia.